Genetics and Rare Diseases Area
Clinical Genetics and Epidemiology
The group has been active since 2005. Our research was originally focused on mitochondrial diseases and later expanded to include a whole series of other genetic disorders.
Currently the group has two main lines of research. The first concerns the biogenesis of the mitochondrial respiratory chain, and in particular of Coenzyme Q and disorders of intermediate metabolism. Our goals are to identify and characterize human genes involved in the biogenesis of the mitochondrial respiratory chain, identify mutations in patients with these diseases, to develop simple models to characterize these mutations, to study their pathophysiology, and test new therapeutic approaches. In recent years we have expanded our scope of research to include other types of metabolic diseases, as well as genetic disorders unrelated to cellular metabolism.
The second field of research concerns genetic diseases in general. The expansion of our diagnostic service has provided us with an incredible amount of genetic data (we have analyzed more than 10,000 patients with NGS, for a variety of different conditions). The main limitation of this approach is that it is often very difficult to establish the pathogenicity of variants identified by diagnostic tests. In recent years, we have developed several models (hybrid minigenes, yeast, CRISPR-CAS9-edited human cells, and the nematode C. elegans) that have allowed us to validate many new variants and establish genotype-phenotypic correlates for several diseases and to identify new genes associated with human diseases.
The main results obtained in the last year are the identification of a specific role for frataxin, the deficient protein in Friedreich’s Ataxia, in the biogenesis of complex I of the respiratory chain, and the identification of the oxidative decarboxylase that catalyzes the first step of the biosynthesis of coenzyme Q ring, an enzyme that has eluded researchers for over 30 years.
Group Members
Prof. Leonardo Salviati – Principal Investigator
Dr. Maria Andrea Desbats – Senior Scientist
Dr. Cinzia Bertolin – Senior Scientist
Dr. Francesca Boaretto – Senior Scientist
Dr. Alessandra Friso – Senior Scientist
Dr. Monica Forzan – Senior Scientist
Dr. Chiara Rigon – Senior Scientist
Dr. Sara Zanchetti – Senior Scientist
Dr. Giuseppe Castello – Senior Scientist
Prof. Matteo Cassina – Senior Scientist
Dr. Carlotta Liccardi – Senior Technologist/Research Associates
Dr. Carmen Manolio – Senior Technologist/Research Associates
Dr. Sofia Tessarolo – Senior Technologist/Research Associates
Dr. Elena Fabiani – Senior Technologist/Research Associates
Dr. Addolorata Cordella – Senior Technologist/Research Associates
Dr. Elisa Baschiera – Post Doc
Dr. Cristina Calderan – Post Doc
Dr. Marco Marchi – Post Doc
Dr. Agata Valentino – PhD Student
Selected Publications
• Calderan C, Sorrentino U, Persano L, Trevisson E, Sartori G, Salviati L, Desbats MA. A yeast based assay establishes the pathogenicity of novel missense ACTA2 variants associated with aortic aneurysms. Eur J Hum Genet. 2024 Mar 15. doi: 10.1038/s41431-024-01591-1.
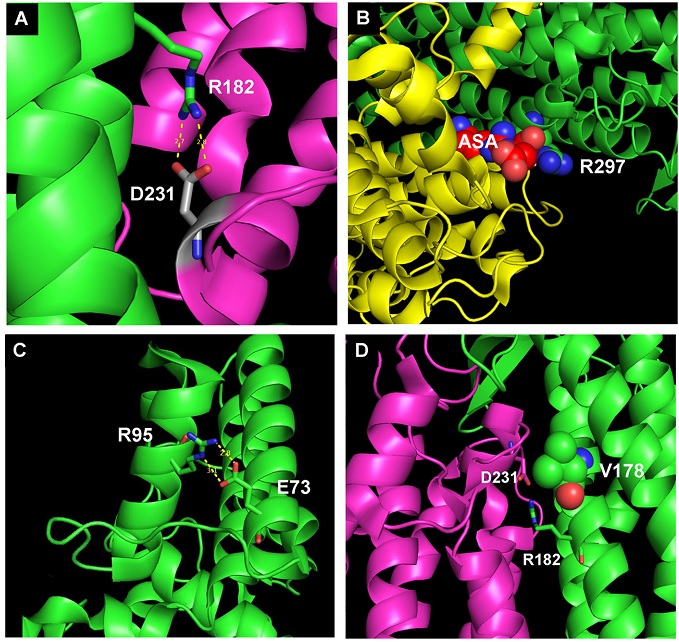
• Pelosi L, Morbiato L, Burgardt A, Tonello F, Bartlett AK, Guerra RM, Ferizhendi KK, Desbats MA, Rascalou B, Marchi M, Vázquez-Fonseca L, Agosto C, Zanotti G, Roger-Margueritat M, Alcázar-Fabra M, García-Corzo L, Sánchez-Cuesta A, Navas P, Brea-Calvo G, Trevisson E, Wendisch VF, Pagliarini DJ, Salviati L, Pierrel F. COQ4 is required for the oxidative decarboxylation of the C1 carbon of coenzyme Q in eukaryotic cells. Mol Cell. 2024 Mar 7;84(5):981-989.e7. doi: 10.1016/j.molcel.2024.01.003.
• Doni D, Cavion F, Bortolus M, Baschiera E, Muccioli S, Tombesi G, d’Ettorre F, Ottaviani D, Marchesan E, Leanza L, Greggio E, Ziviani E, Russo A, Bellin M, Sartori G, Carbonera D, Salviati L, Costantini P. Human frataxin, the Friedreich ataxia deficient protein, interacts with mitochondrial respiratory chain. Cell Death Dis. 2023 Dec 8;14(12):805. doi: 10.1038/s41419-023-06320-y.
• Baschiera E, Sorrentino U, Calderan C, Desbats MA, Salviati L. The multiple roles of coenzyme Q in cellular homeostasis and their relevance for the pathogenesis of coenzyme Q deficiency. Free Radic Biol Med. 2021 Apr;166:277-286.
• Favaro G, Romanello V, Varanita T, Andrea Desbats M, Morbidoni V, Tezze C, Albiero M, Canato M, Gherardi G, De Stefani D, Mammucari C, Blaauw B, Boncompagni S, Protasi F, Reggiani C, Scorrano L, Salviati L, Sandri M. DRP1-mediated mitochondrial shape controls calcium homeostasis and muscle mass. Nat Commun. 2019 Jun 12;10(1):2576.

